KYBELLA® (deoxycholic acid) injection 10 mg/mL
KYBELLA® is the only FDA-approved injectable treatment for adults that destroys fat cells to gradually improve the appearance and profile of moderate to severe fat below the chin (submental fat), also called “double chin.”
ABOUT SUBMENTAL FULLNESS
Submental fullness is an aesthetic condition that may result from the accumulation of subcutaneous fat under the chin. Patients asking about their submental fullness may, at times, refer to it as their “double chin.”
Impacts a broad range of adults, both men and women1,2
Submental fullness may be due to1,2:
- Genetics
- Weight gain
- Aging
Submental fullness is bothersome to many potential patients
1 in 4 adults surveyed online was bothered by submental fullness (N = 4000).4,*
*Online survey of men (51%) and women (49%), ages 18 to 79 years old, who had an annual household income of $50,000 or more.4
The population studied in KYBELLA® clinical trials was 19 to 65 years old
Excess fat below the chin is one of the top 5 aesthetic concerns among aesthetically oriented patients surveyed online.6,†
†Online survey of respondents (N = 7315) who were asked, “How bothered are you by excess fat under the chin/neck?” Respondents were aesthetically oriented (concerned about their appearance and open to aesthetic treatment).
ABOUT KYBELLA®
For adults with moderate to severe submental fat (double chin):
KYBELLA® is an individually tailored* injectable treatment that permanently destroys fat cells in the treatment area under the chin for an improved profile.1
*Multiple injections under the chin per treatment; up to 6 treatments at least 1 month apart.1
KYBELLA® helps you serve a large, diverse patient population as an addition to your core aesthetic portfolio.


CLINICAL STUDY DESIGN
Clinical development program1

2 pivotal clinical studies in North America 2,3
Two randomized, multicenter, double-blind, placebo-controlled trials of identical design were conducted. Subjects received up to 6 treatments with KYBELLA® (N = 514, combined trials) or placebo (N = 508, combined trials) no less than 1 month apart. Subjects ranged in age from 19 to 65 (mean = 49 years), had a BMI ≤ 40 kg/m2 (mean BMI = 29 kg/m2), and represented all Fitzpatrick Skin Types (I-VI). At baseline, 51% of the subjects had a clinician-rated submental fat severity rating of moderate and 49% had a submental fat rating of severe. Efficacy assessments were based on ≥ 2-grade and ≥ 1-grade improvements in submental convexity or fullness on the composite clinician-reported and subject-reported ratings of submental fat 12 weeks after final treatment.
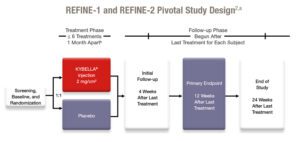
aEnrolling subjects were between ages 19 and 65 with moderate to severe submental fat. Primary endpoints included ≥ 1 -grade and ≥ 2 -grade composite reductions on the Clinician-Reported Submental Fat Rating Scale (CR-SMFRS) and Patient-Reported Submental Fat Rating Scale (PR-SMFRS).
bEfficacy endpoints were based on the change from baseline to 12 weeks after the last treatment.
CLINICAL RESULTS
Key findings from the KYBELLA® pivotal clinical trials include:
≥ 1 – GRADE IMPROVEMENT
Sex: FAge:39BMI (before/after): 19.7/20.4Total treatments: 3Total mLs (all treatment sessions): 12.0
Individual results may vary.
Number of treatments is tailored* to the amount of submental fat and aesthetic goals; 59% of subjects received 6 KYBELLA® treatments in clinical trials.1
*Multiple injections under the chin per treatment; up to 6 treatments at least 1 month apart.1
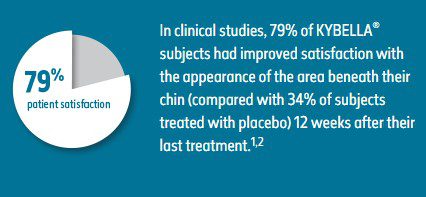
- 68.2% of subjects treated with KYBELLA® were ≥ 1-grade composite responders,† compared with 20.5% of subjects treated with placebo, based on imputed data with the last observation carried forward2
- Individual trial results: ≥ 1-grade composite responders1
— Trial 1: 70.0% KYBELLA® (N = 256) vs 18.6% placebo (N = 250)
— Trial 2: 66.5% KYBELLA® (N = 258) vs 22.2% placebo (N = 258)
≥ 2 – GRADE IMPROVEMENT
Sex: F Age: 34 BMI (before/after): 24.8/25.0 Total treatments: 6 Total mLs (all treatment sessions): 24.0
Individual results may vary.
Number of treatments is tailored* to the amount of submental fat and aesthetic goals; 59% of subjects received 6 KYBELLA® treatments in clinical trials.1
*Multiple injections under the chin per treatment; up to 6 treatments at least 1 month apart.1
- 16.0% of subjects experienced a ≥ 2-grade composite improvement with KYBELLA®,† compared with 1.5% of subjects treated with placebo, based on imputed data with the last observation carried forward2
- Individual trial results: ≥ 2-grade composite responders1
— Trial 1: 13.4% KYBELLA® (N = 256) vs < 0.1% placebo (N = 250)
— Trial 2: 18.6% KYBELLA® (N = 258) vs 3.0% placebo (N = 258)
The most commonly reported adverse reactions in the pivotal clinical trials were injection-site edema/swelling, hematoma/bruising, pain, numbness, erythema, and induration.1
†Based on validated physician and patient measurements.
SAFETY PROFILE
WARNINGS AND PRECAUTIONS1
Marginal Mandibular Nerve Injury
Cases of marginal mandibular nerve injury, manifested as an asymmetric smile or facial muscle weakness, were reported in 4% of subjects in the clinical trials; all cases resolved spontaneously (range 1-298 days, median 44 days). KYBELLA® should not be injected into or in close proximity to the marginal mandibular branch of the facial nerve.
Dysphagia
Dysphagia occurred in 2% of subjects in the clinical trials in the setting of administration-site reactions, eg, pain, swelling, and induration of the submental area; all cases of dysphagia resolved spontaneously (range 1-81 days, median 3 days). Avoid use of KYBELLA® in patients with current or prior history of dysphagia as treatment may exacerbate the condition.
Injection-Site Hematoma/Bruising
In clinical trials, 72% of subjects treated with KYBELLA® experienced hematoma/bruising. KYBELLA® should be used with caution in patients with bleeding abnormalities or who are currently being treated with antiplatelet or anticoagulant therapy as excessive bleeding or bruising in the treatment area may occur.
Risk of Injecting Into or in Proximity to Vulnerable Anatomic Structures
To avoid the potential of tissue damage, KYBELLA® should not be injected into or in close proximity (1 cm-1.5 cm) to salivary glands, lymph nodes, and muscles. Care should be taken to avoid inadvertent injection directly into an artery or a vein as it can result in vascular injury.
Injection Site Alopecia
Cases of injection site alopecia have been reported with administration of KYBELLA®. Onset and duration may vary among individuals and may persist. Consider withholding subsequent treatments until resolution.
Injection Site Ulceration and Necrosis
Injections that are too superficial into the dermis may result in skin ulceration and necrosis. Cases of injection site ulceration and necrosis have been reported with administration of KYBELLA®. Do not administer KYBELLA® into affected area until complete resolution.
ADVERSE REACTIONS1
The most commonly reported adverse reactions in the pivotal clinical trials were: injection site edema/swelling, hematoma/bruising, pain, numbness, erythema, and induration.
Adverse Reactions in the Pooled Trials 1 and 2 1,a
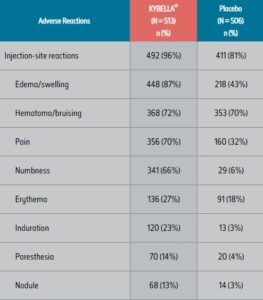
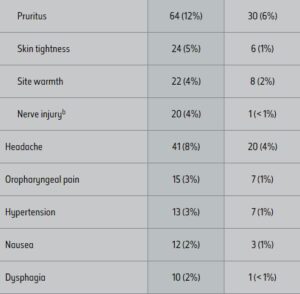
aAdverse reactions that occurred in ≥ 2% of KYBELLA® treated subjects and at greater incidence than placebo.
bMarginal mandibular nerve paresis.
MECHANISM OF ACTION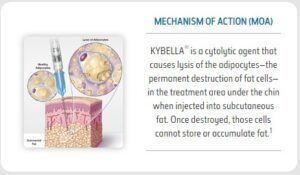
- KYBELLA® is a nonhuman and nonanimal formulation of deoxycholic acid, a cytolytic agent1
- Deoxycholic acid is a naturally occurring molecule in the body that aids in the breakdown and absorption of dietary fat2
- Fat cleared from the treatment area is processed through natural metabolic mechanisms3
Pharmacokinetics of deoxycholic acid from KYBELLA® after injection
Following subcutaneous injection, KYBELLA® is rapidly absorbed into the plasma and enters the body’s bile acid pool in the enterohepatic circulation, where it is then excreted along with the endogenous deoxycholic acid.1 Posttreatment deoxycholic acid plasma levels return to the endogenous range within 24 hours.1 No accumulation is expected with the proposed treatment frequency (minimum 1-month treatment intervals).1
ABOUT TREATMENT
What to know about KYBELLA® dosing and administration:
- KYBELLA® is injected into subcutaneous fat tissue in the submental area using an area-adjusted dose of 2 mg/cm2
— A single treatment consists up to a maximum of 50 injections, 0.2 mL each (up to a total of 10 mL), spaced 1 cm apart1
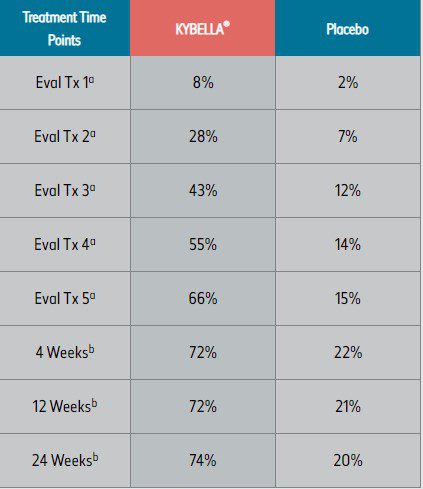
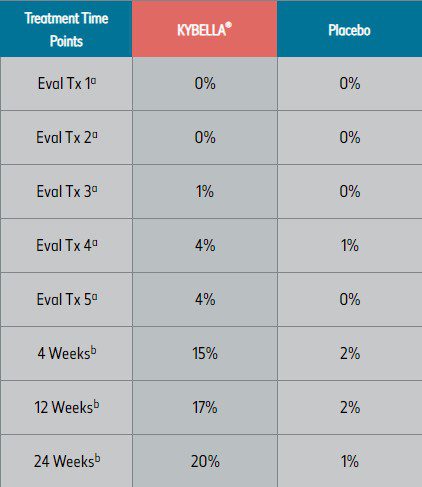
— Up to 6 single treatments may be administered at intervals no less than 1 month apart1 - In clinical studies, many KYBELLA® subjects experienced visible results in 2 to 4 treatment sessions (see tables below)2
— 59% of subjects received 6 KYBELLA® treatments in clinical trials1
Pivotal clinical trials2
aTreatment (Tx) evaluation time points.
bWeeks after final treatment.
THE INJECTION PROCESS MAY TAKE 15 to 20 MINUTES
- In clinical trials, subjects were administered an average of 6.4 mL at the first treatment session, and subjects who received all 6 treatments were administered an average of 4.4 mL at the sixth treatment session; no more than 10 mL per treatment session may be administered1
- The safe and effective use of KYBELLA® depends on the use of the correct number of and locations for injections, proper needle placement, and proper administration techniques1
- Healthcare professionals administering KYBELLA® must understand the relevant submental anatomy and associated neuromuscular structures in the area involved, as well as any alterations to the anatomy due to prior surgical or aesthetic procedures1
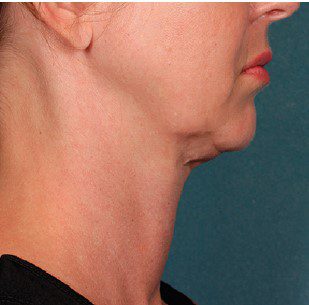
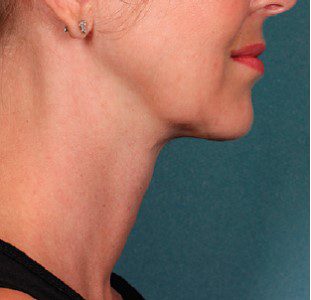
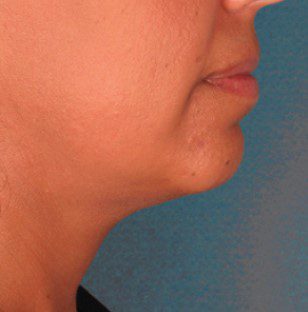
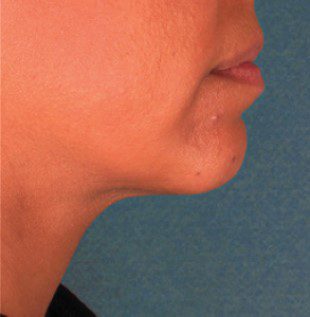
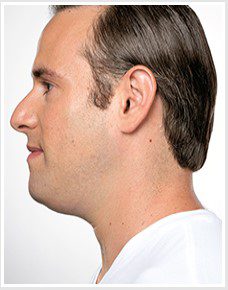
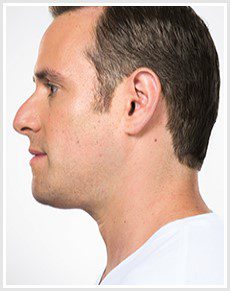
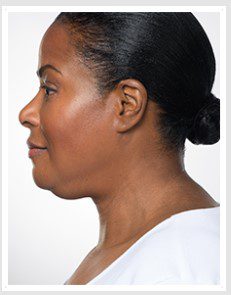
BEFORE & AFTER
KYBELLA® is an individually tailored* injectable treatment for adults with moderate to severe submental fat (double chin).1 The number of treatments is tailored* to the amount of submental fat and aesthetic goals of each patient; 59% of subjects received 6 KYBELLA® treatments in clinical trials.1
*Multiple injections under the chin per treatment; up to 6 treatments at least 1 month apart.1
The safe and effective use of KYBELLA® for the treatment of subcutaneous fat outside the submental area has not been established and is not recommended.1
Not all patients are candidates for KYBELLA® treatment:
- KYBELLA® is contraindicated in the presence of infection at the injection sites1
- Give careful consideration to patients with prominent platysmal bands or excessive skin laxity, who may not achieve aesthetically desirable results1
- Avoid use of KYBELLA® in patients who have a current or prior history of dysphagia or cause of submental fullness other than submental fat (eg, thyroid abnormality or cervical lymphadenopathy)1
- Use caution in patients who have had prior surgical or aesthetic treatment of the submental area, and in patients with bleeding abnormalities or who are currently being treated with antiplatelet or anticoagulant therapy1
Unretouched photos of paid model.
Sex: M Age: 36 BMI (before/after): 26.6/26.3
Total treatments: 6 Total mLs (all treatment sessions): 28.0
Individual results may vary.
Unretouched photos of paid model.
Sex: F Age: 48 BMI (before/after): 29.1/29.1
Total treatments: 6 Total mLs (all treatment sessions): 23.4
Individual results may vary.
INDICATION
KYBELLA® (deoxycholic acid) injection is indicated for improvement in the appearance of moderate to severe convexity or fullness associated with submental fat in adults.
The safe and effective use of KYBELLA® for the treatment of subcutaneous fat outside the submental region has not been established and is not recommended.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
KYBELLA® is contraindicated in the presence of infection at the injection sites.
WARNINGS AND PRECAUTIONS
Marginal Mandibular Nerve Injury
Cases of marginal mandibular nerve injury, manifested as an asymmetric smile or facial muscle weakness, were reported in 4% of subjects in the clinical trials; all cases resolved spontaneously (range 1-298 days, median 44 days). KYBELLA® should not be injected into or in close proximity to the marginal mandibular branch of the facial nerve.
Dysphagia
Dysphagia occurred in 2% of subjects in the clinical trials in the setting of administration-site reactions, eg, pain, swelling, and induration of the submental area; all cases of dysphagia resolved spontaneously (range 1-81 days, median 3 days). Avoid use of KYBELLA® in patients with current or prior history of dysphagia as treatment may exacerbate the condition.
Injection Site Hematoma/Bruising
In clinical trials, 72% of subjects treated with KYBELLA® experienced hematoma/bruising. KYBELLA® should be used with caution in patients with bleeding abnormalities or who are currently being treated with antiplatelet or anticoagulant therapy as excessive bleeding or bruising in the treatment area may occur.
Risk of Injecting Into or in Proximity to Vulnerable Anatomic Structures
To avoid the potential of tissue damage, KYBELLA® should not be injected into or in close proximity (1 cm-1.5 cm) to salivary glands, lymph nodes, and muscles. Care should be taken to avoid inadvertent injection directly into an artery or a vein as it can result in vascular injury.
Injection Site Alopecia
Cases of injection site alopecia have been reported with administration of KYBELLA®. Onset and duration may vary among individuals and may persist. Consider withholding subsequent treatments until resolution.
Injection Site Ulceration, Necrosis, and Infection
Injections that are too superficial into the dermis may result in skin ulceration and necrosis. Cases of injection site ulceration, necrosis, and infection have been reported with administration of KYBELLA®. Some cases of injection site infection have included cellulitis and abscess requiring antibiotic treatment and incision and drainage. Do not administer KYBELLA® into affected area until complete resolution.
ADVERSE REACTIONS
The most commonly reported adverse reactions in the pivotal clinical trials were: injection site edema/swelling, hematoma/bruising, pain, numbness, erythema, and induration.
Please see KYBELLA® full Prescribing Information.
References: 1. KYBELLA® Prescribing Information, October 2022. 2. Data on file, Allergan, April 2015; MD Treatments Among Men.