Single Dose Vial
-
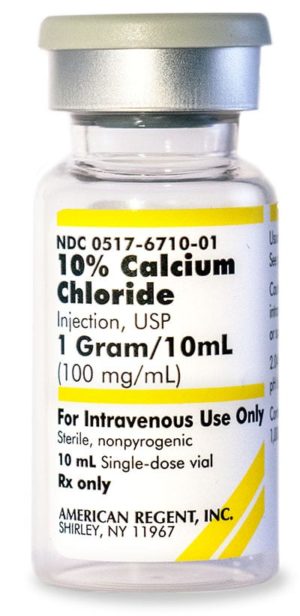
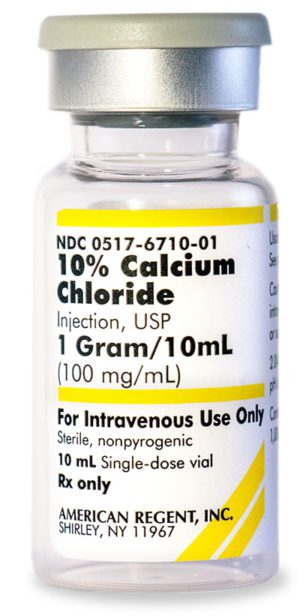
10% Calcium Chloride Injection, USP 028867198000
NDC Number 0517-6710-10
NDC Number 00517671010
Manufacturer: American Regent
Application Replacement Preparation
Strength: 100mg/mL
Single Dose Vial
Storage Requirements USP Controlled Room Temperature
Type Intravenous
Volume 10 mLPrice range: $33.99 through $339.90 -
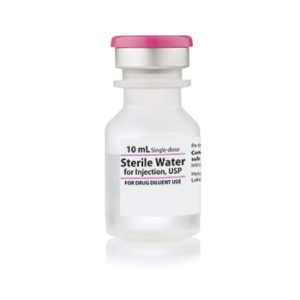
Sterile Water for Injection
Sterile Water for Injection, Preservative Free USP
Application: Diluent
Single Dose Fliptop Vial
Non-DEHP
Preservative Free
Latex Free: Latex Information: Natural rubber latex has not been used in the manufacture of this device or drug container closure system
Manufacturer: HospiraPrice range: $15.99 through $326.99 -
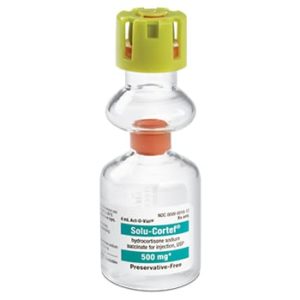
Solu-Cortef
Manufacturer: Pfizer
Brand Solu-Cortef®
Generic Drug Name Hydrocortisone Sodium Succinate, Preservative Free
Application Anti-inflammatory Agent
Single Dose Vial
Storage Requirements USP Controlled Room Temperature
Type Intramuscular or IntravenousPrice range: $30.99 through $113.99 -
Ropivacaine HCl, Preservative Free 5mg/ml 150mg/30ml 1 x 10 NDC 032221791375
Ropivacaine HCl, Preservative Free5mg/ml 150mg/30ml 1 x 10 (Box of 10 Vials)
Infiltration, Nerve Block and Epidural Injection Single Dose Vial 10 mL
NDC# 0409-9301-30 / 00409930130 / 0409930130Non-Returnable
$104.99 -
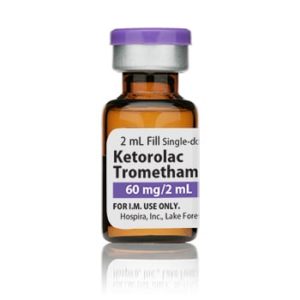
Ketorolac Tromethamine Injection
Manufacturer: Hospira
Application Nonsteroidal Anti-inflammatory Agent
Strength: 30mg/mL
Generic Ketorolac Tromethamine, Preservative Free
Single Dose Vial
Storage Requirements USP Controlled Room Temperature
Latex Information indicates that natural rubber latex has not been used in the manufacture of this device or drug container closure systemPrice range: $49.99 through $54.99 -
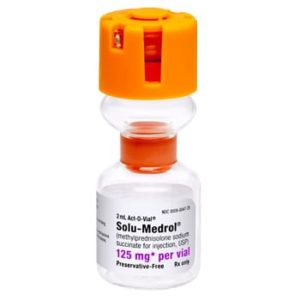
Solu-Medrol Methylprednisolone Sodium Succinate 032336805000
NDC Number 00009-0047-22 / 0009004722
Manufacturer Pfizer
Brand Solu-Medrol®
Application Corticosteroid
Strength: 125mg/2mL
Generic Equivalent to Marcaine®, Sensorcaine®
Single Dose Vial – Act-O-Vial
Storage Requirements USP Controlled Room Temperature
Volume 2 mL$14.99 -
Solu-Medrol Methylprednisolone Sodium Succinate 40MG 032365935000
NDC Number 00009-00039-28 / 00009003928
Brand Solu-Medrol®
Manufacturer: Pfizer
Application Corticosteroid
Strength: 40mg/mL
Generic Equivalent to Methylprednisolone Sodium Succinate, Preservative Free
Single Dose Vial
Storage Requirements USP Controlled Room Temperature
Type Intramuscular, Intravenous, or Subcutaneous
Volume 1 mL$9.99 -
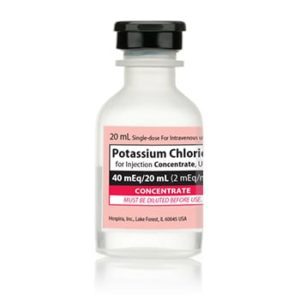
Potassium Chloride Concentrate 030827238000
NDC Number 00409-6653-05 / 00409665305
Manufacturer: Hospira
Application Replacement Preparation
Strength: 2 mEq/mL
Generic Equivalent to Marcaine®, Sensorcaine®
Single Dose Vial
Storage Requirements USP Controlled Room Temperature
Type Intravenous
Volume 20 mL
Latex Information indicates that natural rubber latex has not been used in the manufacture of this device or drug container closure system$9.50