injection
-
FOUNDATIONS OF AESTHETICS FOR DENTISTS | Hands-On Course
The Foundations of Advanced Aesthetics course now has a specific component just for Dentists! We have listened to your requests and developed a curriculum that provides you with an all-encompassing knowledge of lower facial rejuvenation!
Skill Level:
BEGINNER/INTERMEDIATE
Virtual Observation & Hands on training options available.
HANDS ON
Includes Hands-On, One-On-One training (worth 5.5 CME credits) and on-demand didactic course (10 CME credits).
VIRTUAL OBSERVATION + DIDACTIC
These dates include the Virtual Observation portion ONLY. CME credits are NOT awarded for these dates.
VIRTUAL OBSERVATION
These dates include the Virtual Observation portion ONLY. CME credits are NOT awarded for these dates.
DIDACTIC ONLY (10 CME CREDITS)
Our virtual didactic offers students a premier training experience, 100% online. you will get access to our 10-Hours of courses that cover topics like anatomy, product safety, patient assessments, product rheology, and much more. Students will be given access to didactic courses for 1 year from purchase date.
$2,997.00 -
Sterile Water for Injection
Sterile Water for Injection, Preservative Free USP
Application: Diluent
Single Dose Fliptop Vial
Non-DEHP
Preservative Free
Latex Free: Latex Information: Natural rubber latex has not been used in the manufacture of this device or drug container closure system
Manufacturer: HospiraPrice range: $15.99 through $326.99 -
LIP ASSESSMENT & INJECTION | Webinar
Join Lori Robertson, MSN, FNP-C for this one-hour webinar on assessing and injecting lips.
2019 | TREATMENT AREA: LIPS | PROCEDURE: SOFT TISSUE FILLERS
Overview
Injection of Lips: Lori Robertson, MSN, FNP-C.
Highlights:
- General principles and practical tips for assessing and injecting lips
- Lip changes through aging
- Surface anatomical landmarks and location of critical arteries
- Proper injection techniques and methods
- Review of useful and safe lip filler choice
- Post-treatment recommendations to minimize adverse outcomes and potential complications
Categories
Instructor Procedure Area of Treatment $199.00 -
Non-Vented Dispensing Pin with SAFSITE Valve with Luer Lock Connector 034173982500
Automatic two-way valve for aspiration or injection of medication from inverted bags or semi-rigid plastic containers.
Product Identification
Product Code DP3500L
NDC Number 08021-4135-01
Reference NumberSafety Data
BPA Information Contains BPA
DEHP Information Components Do Not Contain DEHP
Latex Information Components Do Not Contain Natural Rubber
PVC Information Components Do Not Contain PVCPrice range: $3.25 through $155.00
Non-Vented Dispensing Pin with SAFSITE Valve with Luer Lock Connector 034173982500
Price range: $3.25 through $155.00 Select options -
CYANOCOBALAMIN PACKAGE
SUPER B-12 PACKAGE WITH CYANOCOBALMIN 30ML (COBAL 1000) INJECTION, USP, MDV, 1000MCG, NDC # 67457-400-05
Package Items Include:
5 x 30mL Cyanocobalamin MDV
Choice of One Box:
_____ Insulin Syringes 1cc 30G x 5/16, Bx/100
_____ Insulin Syringes 1/3cc 31 x 5/16, Bx/100
_____3cc 23G x 1”, Bx/100 #26101
_____3cc 25G x 5/8, Bx/100 #26100
_____3cc 25G x 1”, Bx/100 #044327777
Included:
_____Alcohol Prep Pads, Medium, Bx/200
_____Sharps Container, Red, 2 Gallon
_____ Band-aids, Bx/100, 1” x 3” or Spots
$150.00 -
0.2 Micron SUPOR Aspiration / Injection Disc Filter 032511398437
Braun # 415002, 0.2 Micron Supor Aspiration/ Injection Disc Filter
Proximal and distal Luer lock connections designed for bacteria retentive filtration of medication
Fluid retention: 0.3 mL
Color: Green
Product Identification
Product Code PF2000
NDC Number 08021-4150-02
Reference Number 415002
Safety Data
Latex Information Components Do Not Contain Natural RubberPrice range: $3.11 through $137.50
0.2 Micron SUPOR Aspiration / Injection Disc Filter 032511398437
Price range: $3.11 through $137.50 Select options -
B Braun IV Solutions Sterile Water Injection USP B Braun IV Solutions
Generic Drug Name Sterile Water for Injection, Preservative Free
Manufacturer # L8501-01
Manufacturer B. Braun
Application Diluent
Container Type Flexible Bag
Dosage Form IV Solution
NDC Number 00264-7850-10
Type Intravenous
Size Volume 500 mLNot manufactured with latex, PVC or DEHP.
Price range: $7.00 through $127.00
B Braun IV Solutions Sterile Water Injection USP B Braun IV Solutions
Price range: $7.00 through $127.00 Select options -
Ketorolac Tromethamine Injection
Manufacturer: Hospira
Application Nonsteroidal Anti-inflammatory Agent
Strength: 30mg/mL
Generic Ketorolac Tromethamine, Preservative Free
Single Dose Vial
Storage Requirements USP Controlled Room Temperature
Latex Information indicates that natural rubber latex has not been used in the manufacture of this device or drug container closure systemPrice range: $49.99 through $54.99 -
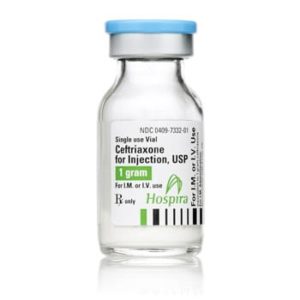
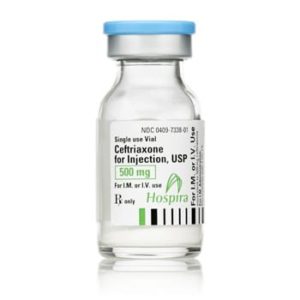
Ceftriaxone for Injection
Ceftriaxone for Injection, USP
Brand Name Equivalent: Rocephin®
Manufacturer Hospira/Pfizer
15mL Single Dose Vial“Latex Information” indicates that natural rubber latex has not been used in the manufacture of this device or drug container closure system.
“Preservative Information” indicates that this product does not contain ingredients identified as a preservative, as defined by the USP.
Price range: $21.00 through $53.00 -
Plenamine 15% Amino Acids Injection 1000 mL 026346274750
Latex Free, DEHP Free BAG
Per the manufacturer: Acceptable Dating: >= 90 days
Pharmacy Bulk Package is a sterile, clear, nonpyrogenic solution of essential and nonessential amino acids for intravenous infusion in parenteral nutrition following appropriate dilution.
Plenamine™ 15% in a Pharmacy Bulk Package is not for direct infusion. It is a sterile dosage form which contains several single doses for use in a pharmacy admixture program in the preparation of intravenous parenteral fluids.
Non-Returnable.
$51.99 -
Aesthetic Needle
100/Box
Price range: $20.99 through $51.98 -
Manganese Chloride Injection USP, Preservative Free 0.1 mg / mL 10 mL 03150675000
NDC Number 00409-4091-01 / 00409409101
Manufacturer: Hospira
Application Trace Element
Strength: 0.1mf/mL
Generic Equivalent to Marcaine®, Sensorcaine®
Single Dose Vial
Storage Requirements USP Controlled Room Temperature
Type Intravenous
Volume 10 mL
Latex Information indicates that natural rubber latex has not been used in the manufacture of this device or drug container closure system$46.99 -
Dexamethasone Sodium Phosphate 029643576000
Dexamethasone Sodium Phosphate 4 mg / mL Intramuscular or Intravenous Injection Multiple Dose Vial 30 mL
NDC Number 6745742130 / 67457-421-30
Manufacturer: Mylan Institutional
Application Corticosteroid
Strength: 4mg/mL
Multiple Dose Vial
Generic Name Dexamethasone Sodium Phosphate
Storage Requirements USP Controlled Room Temperature
Type Intramuscular or Intravenous / Injection
Volume 30 mL
Latex Information indicates that natural rubber latex has not been used in the manufacture of this device or drug container closure system$23.70 -
Solu-Medrol Methylprednisolone Sodium Succinate 032336805000
NDC Number 00009-0047-22 / 0009004722
Manufacturer Pfizer
Brand Solu-Medrol®
Application Corticosteroid
Strength: 125mg/2mL
Generic Equivalent to Marcaine®, Sensorcaine®
Single Dose Vial – Act-O-Vial
Storage Requirements USP Controlled Room Temperature
Volume 2 mL$14.99 -
Solu-Medrol Methylprednisolone Sodium Succinate 40MG 032365935000
NDC Number 00009-00039-28 / 00009003928
Brand Solu-Medrol®
Manufacturer: Pfizer
Application Corticosteroid
Strength: 40mg/mL
Generic Equivalent to Methylprednisolone Sodium Succinate, Preservative Free
Single Dose Vial
Storage Requirements USP Controlled Room Temperature
Type Intramuscular, Intravenous, or Subcutaneous
Volume 1 mL$9.99 -
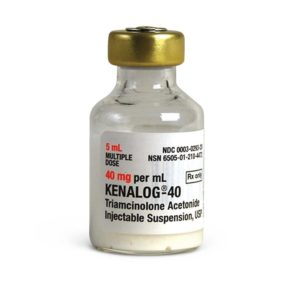
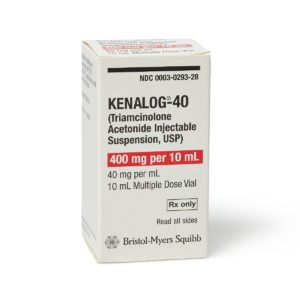
Kenalog
Manufacturer: Bristol-Myers Squibb
Brand Kenalog®-40
Application Corticosteroid
Generic Equivalent to Marcaine®, Sensorcaine®
Multiple Dose Vial
Storage Requirements USP Controlled Room Temperature
Type Injectable / Injection / Intramuscular or Intra-Asticular$65.00