- Medspa Essentials
- Aestheticians
- Apparel
- Clinic Furniture
- Devices
- Pharmaceutical & Medical Supplies
- Safety Supplies
- Training & Services
- Vendors
- Acara Partners
- Aesthetic Record
- Aesthetic Next
- American Med Spa Association
- CEDR HR Solutions
- Cervello & Co.
- Colorescience
- CosmoGlo
- Docovia
- Eden Skin And Body
- Endless Attraction
- FACE Med Store
- Healexir
- KA Ring Designs
- LIFTIE Aspirator
- Massage Tools
- Merit Pharmaceutical
- National Medical Directors
- Post Love Skincare
- Rana Kennelley
- Renee John Uniforms
- Source One Beauty
- SyringeRack
- The Aesthetic Immersion
- Velez by Vesna
- We Treat
Flash sale
Fresh deal everyday. Get it before time runs out
-
$58.99
$69.99Disposable Fabric Pillow Covers – 21″ x 23″ – 100PK (5 x 20PK)
Sold By: Source One Beauty -
$2,699.00
$3,099.00Spa Numa NEW MEDICI Heavy Duty Medical Grade Pedestal 4-Motor Treatment Chair
Sold By: Source One Beauty -
$2,599.00
$2,899.00Spa Numa SERENITY Electric 4 Motor Treatment Chair for Aesthetic & Medical Procedures
Sold By: Source One Beauty
Product Categories
Show MoreShow Less
Filter by price
Color
Size
Edit Content
Product Categories
Show MoreShow Less
Filter by price
Color
Size
-
Strep A Test Kit QuickVue In-Line Strep A Test Kit – 25 tests per box Quidel
Sold By: Merit PharmaceuticalThe QuickVue In-Line Strep A test detects group A streptococcal antigen directly from a throat swab.
The test is an easy one-step process. Results are available in 5 minutes, and are presented in two colors for easy interpreting. The exclusive in-line extraction and rapid procedure allow testing to take place at point of care. Patients can start treatment immediately resulting in better patient care.
The QuickVue has an overall accuracy of 96%. The kit includes external controls so no additional ordering is necessary. The test is CLIA waived.
The kit includes:
Individually packaged test cassettes (25)
Extraction solution bottles (25)
4M Sodium Nitrite (0.38 mL), and 0.2M Acetic Acid (0.43 mL) inside glass ampule
Individually packaged sterile rayon-tipped swabs on green shafts (25)
Positive control swab (+) (1)
Negative control swab (–) (1)
Extraction kit (1)
5 tubes
5 disposable droppers
Package insert (1)
Procedure card (1)$103.00 -
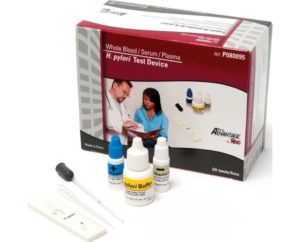
H. Pylori Test Device 20/Bx 012898152462
Sold By: Merit PharmaceuticalH. Pylori Test Device Kit, 20 Tubes
One-step, qualitative membrane strip-based immunoassay designed for detection of H. pylori IgG antibodies in whole blood, serum or plasma
Test results in 10 minutes helps assist physicians in making treatment decisions during office visit
88% sensitivity with 89% specificity
CLIA waived for whole bloodIncludes:
- 20 Individually Packaged H.pylori Test Devices
- Disposable Sample Droppers
- 20 Disposable Heparinized Capillary Tubes & Dispensing Bulk
- 1 Positive Control
- 1 Negative Control
- 1 Sample Buffer
- 20 test/kit
$105.00 -
BD Vacutainer Safety-Lok Blood Collection Set
Sold By: Merit PharmaceuticalManufacturer Becton Dickinson
Winged Blood Collection Set With Luer Adapter SAFETY
Tubing Length 12 Inch Tubing$113.00 – $127.50Sold By: Merit PharmaceuticalBD Vacutainer Safety-Lok Blood Collection Set
$113.00 – $127.50 Select options -
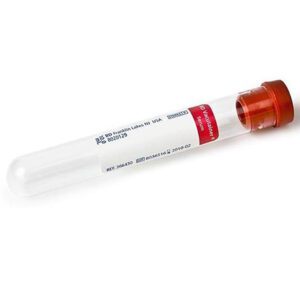
BD 366430 Vacutainer Serum Blood Collection Tubes 10 mL Glass 16mm x 100mm, 100/box
Sold By: Endless AttractionBD 366430 Vacutainer Serum Blood Collection Tubes 10 mL Glass 16mm x 100mm. BD Vacutainer Plus Plastic Serum Tubes have spray-coated silica and are used for serum determinations in chemistry. Samples processed in these tubes may also be used for routine blood donor screening, immunohematology and diagnostic testing of serum for infectious disease.
$129.00$135.00 -
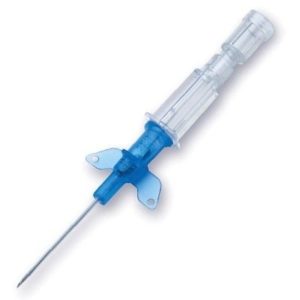
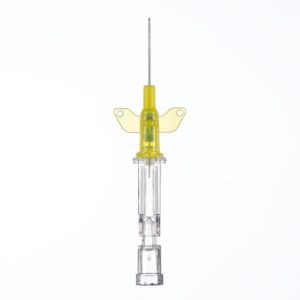
Introcan Safety IV Catheter Winged
Sold By: Merit PharmaceuticalIntrocan Safety®
Manufacturer B. Braun
Plastic Hub Material
WINGED Hub / Winged Catheter
Stainless Steel Needle
Universal Bevel Needle Point Style
Pressure Rating 300 psi
X-Ray Compatibility RadiopaqueBOX/50
$152.60 – $165.99 -
Orasure Quickflu Rapid Flu A & B Test, 22 Tests Box 013602705375
Sold By: Merit PharmaceuticalOrasure Quickflu Rapid Flu A & B Test, 22 Tests/Bx
# 1001-0320
Non-ReturnableThe OraQuick® HCV test is FDA approved for detecting HCV antibodies in fingerstick and venipuncture whole blood. Our simple platform enables healthcare providers to deliver an accurate diagnosis in 20 minutes.
SENSITIVITY TO MEET YOUR NEEDS
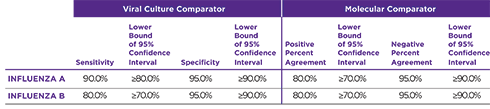
OraSure QuickFlu® is an FDA cleared, CLIA-waived in vitro rapid qualitative test for the detection and differentiation of influenza type A and type B. An easy nasal swab procedure provides accurate results in as little as 10 minutes.
Orasure QuickFlu® delivers highly accurate results for flu types A and B and meets FDA’s new standards of performance.
- Proven analytical sensitivity across 51 different flu strains including CDC’s 2019-2020 flu strain panels
- Exceptional correlation to culture/PCR
PROVEN PERFORMANCE THAT MEETS FDA’S RECLASSIFICATION RULES
OraSure QuickFlu® exceeds the FDA’s reclassification requirement minimums for RIDTs.
- As of February 13, 2017, all RIDTs must meet the minimum acceptance performance criteria and comparator method for antigen based RIDTs. Tests that do not meet the new requirements cannot be used.
- Analytical reactivity testing must be performed on contemporary influenza strains and newly emerging strains in the event of an emergency.
ENSURES RESPONSIVE STANDARD OF CARE
-
- Simple, easy-to-use procedure with built-in control line
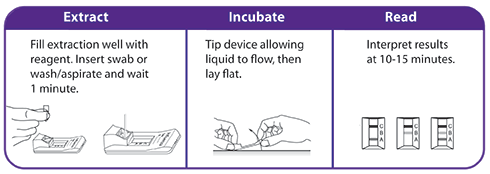
- Includes a flocked swab that optimizes sample collection and release resulting in superior performance
- American Society of Microbiology says, “NP flocked swabs collect better samples than routine NP swabs”
- Nasal swab collection – comfortable for the patient and simple for the HCP with no ancillary supplies required
- Ready-to-use reagents with room temperature storage
- Rapid testing that maximizes results
TEST AT THE POINT OF CARE
OraSure QuickFlu® is ideal for testing at the point of care in…
- Laboratories
- Emergency Rooms
- Physician Offices
- Public Health Facilities and Clinics
REIMBURSEMENT INFORMATION
CPT Codes
87804 QW- Influenza A Detection
87804-59- Influenza B Detection
$318.99 -
OraQuick Advance HIV Rapid Test Kit
Sold By: Merit PharmaceuticalOraQuick Advance® HIV Rapid Test Kit:
20 minute Results
CLIA Waived
25 Test Count Packaging Includes: (25) Divided Pouches w/ (1) Test Device, (1) 1mL Vial Developer Solution, (5) Reusable Test Stands, (25 Specimen Collection Loops, (25) Information Pamphlets, Package Insert, Customer Letter
# 1001-0079 1 X 25TESTNOTE: LEAD TIME TO RECEIVE THE TEST IS ONE – TWO WEEKS.
$430.00 -
OraQuick HCV Rapid Test Kit 25 Test Kits (Hepatitis C)
Sold By: Merit PharmaceuticalOraQuick® HCV Rapid Test Kit:
20 minute Results
CLIA Waived
25 Test Count Packaging Includes: (25) Divided Pouches Each Containing a Test Device, Absorbent Packet, Developer Solution Vial (each contains 0.75mL of a Buffered Saline Solution w/ an Antimicrobial Agent), (5) Reusable Test Stands, (25), Specimen Collection Loops, Package Inserted
NOTE: LEAD TIME TO RECEIVE THE TEST IS ONE – TWO WEEKS.
$509.99